It is well known that the performance of production equipment in the biopharmaceutical industry has a direct impact on product quality and even on the capacity of the entire industry. Stainless steel equipment and disposable equipment are now widely used in biopharmaceutical companies, but what are the applicable scenarios for these two types of equipment? How to use them or even use them in combination to bring clients maximum economic benefits and industrialize new technologies and products in faster steps?
As a benchmark company providing core equipment, process systems and engineering solutions in China, Morimatsu provide stainless steel, disposable or mixed solutions in line with the actual needs of clients. The following is our brief description of the strengths and weaknesses of stainless steel equipment and disposable equipment used in the production of biopharmaceuticals.
The typical stainless steel system facilitates mass production due to the high degree of automation and process stability. All electronic data and other information are collected easily, and batch records can be generated automatically, basically eliminating the need for operator tracking. And there is a low operating cost, and the equipment can be reused after cleaning and disinfection to reduce solid waste. However, stainless steel equipment also has the weaknesses of high initial investment, long construction period, large utility consumption, and the need for more cleaning validation. Usually, clients in late clinical stage or requiring mass production give priority to stainless steel equipment. Moreover, since there is usually no need to consider consumables for stainless steel equipment, it has a fairly significant cost advantage in automated mass production.




Stainless steel biopharmaceutical system delivered by Morimatsu
Besides, with the change of national macro strategy from “generic drug” to “innovative drug” and the introduction of MAH and other policies, a large number of R&D-oriented enterprises have emerged in China. With its advantages of less need for cleaning validation, low initial investment, short construction period, small utility consumption and less plant footprint, the disposable production equipment is favored by R&D-oriented enterprises, small-scale manufacturers and some CDMO companies. Inevitably, however, disposable products are also facing multiple challenges. The use of disposable equipment leads to an increase in manual operations, resulting in more pain points in production management and a more complex validation process for the product at a later stage. Moreover, the supply chain of disposable equipment is always facing the challenge of instability due to the higher cost of consumables and the production of more solid waste. For example, for disposable bioreactors, there are only a few global supplies of equipment larger than 2000L in size, which limits the engagement of some enterprises in rapid mass production for some products.


Disposable products delivered by Morimatsu
The following are four combination strategies that can be adopted by upstream and downstream systems of an antibody project.
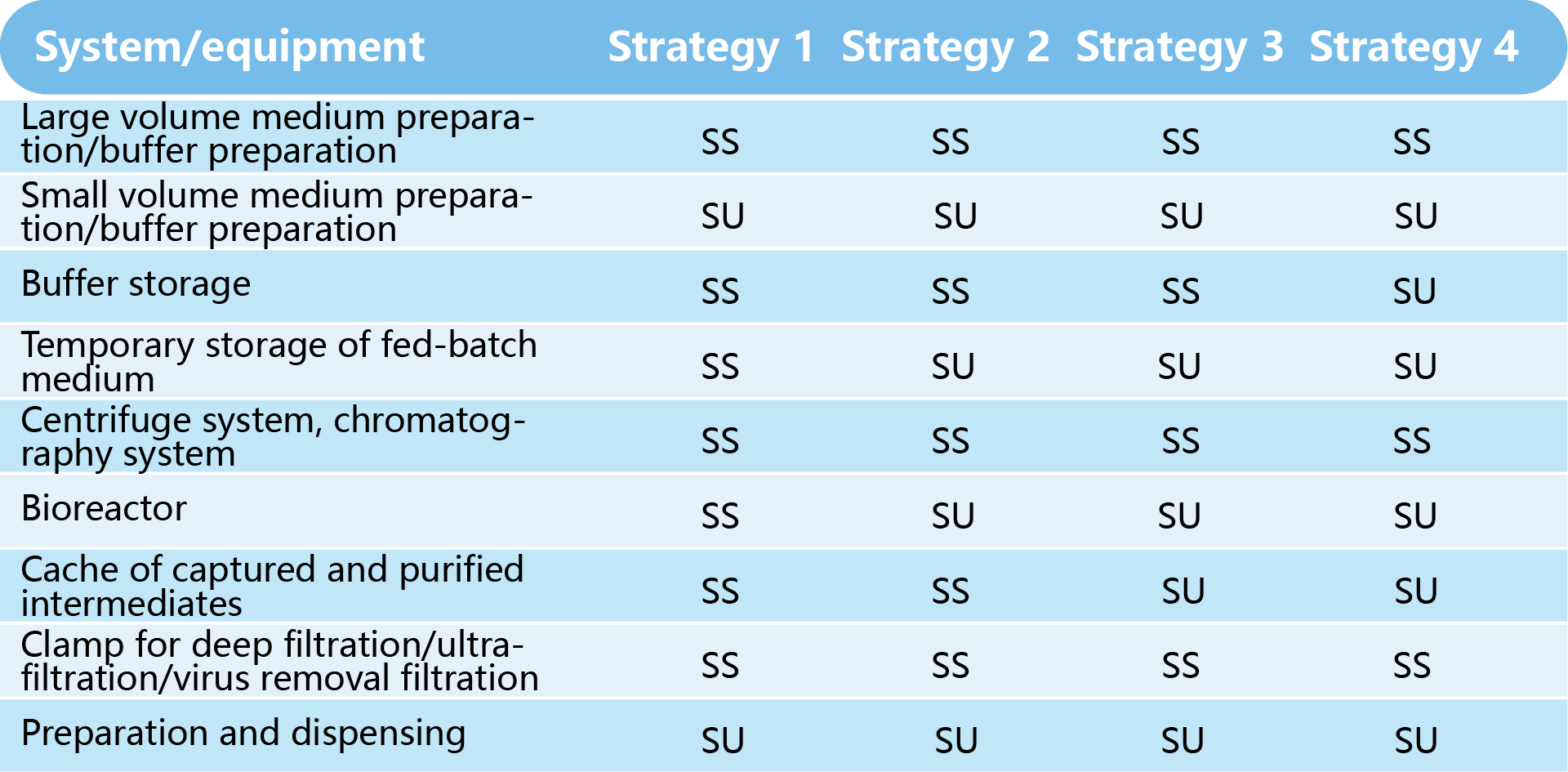
SS: stainless steel system; SU: disposable system
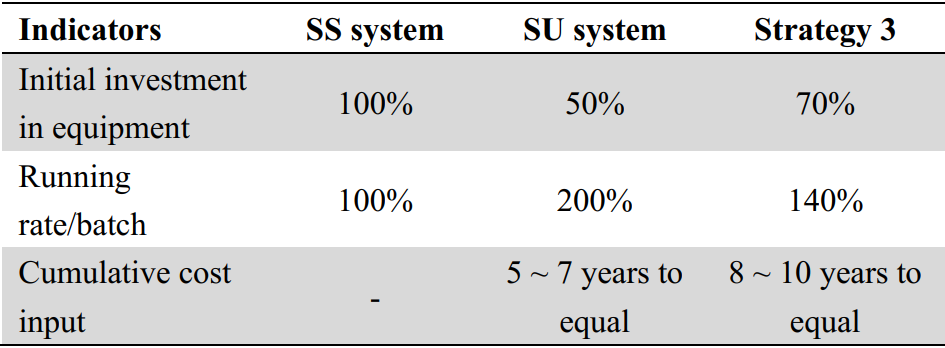
Assume that the output of this antibody is 40 batches/year, the expression quantity is 5g/L, the facilities are two 2000L upstream systems and a matching downstream line, the entire system is made of stainless steel, the initial investment cost is 100%, and the operating cost of each batch is also 100% for comparative analysis as a benchmark, the initial cost will be at a low level of about 50% if fully equipped with disposable equipment, but the operating cost will rise to about 200%. The total cost input will equal the investment in stainless steel equipment in 5 ~ 7 years. And when the plant runs for more than 5~7 years, the cumulative cost will be higher than that of the plant with stainless steel system. By analyzing all factors, Morimatsu can offer customers a solution that mixes stainless steel and disposable systems, with an initial investment of about 70% and an operating cost of about 140% of full use of stainless steel system. It offers certain advantages on the whole, such as low initial investment, increased flexibility in local areas, and a delay of 8 to 10 years for cumulative cost input to equal investment in stainless steel equipment.
The specific mixed strategy to be used should take into account the production scale of companies, the expected speed to marketization, the type of product, the use period of the product, the initial cost and the operating habits. Morimatsu will assist clients in putting their actual needs into practice and offer a wide range of solutions in stainless steel, disposable or hybrid versions.
Morimatsu will continue to focus on the manufacturing and R&D of core equipment, process systems and engineering solutions in the future, with continuous efforts to pursue higher quality and higher added value, boost the development of the industry and contribute to a healthier, more beautiful and more convenient life for the public.
About Shanghai Morimatsu Pharmaceutical Equipment Engineering Co., Ltd.
Shanghai Morimatsu Pharmaceutical Equipment Engineering Co., Ltd (Stock Code: 2155.HK) is a wholly-owned subsidiary of Morimatsu International Holdings Company Limited. Morimatsu, founded in Japan and taken root in China, has developed into a multi-national company that embraces globalization, masters core technology, and gains rich experience from project implementation in diverse fields including core equipment, process systems, and engineering solutions. The company has built advanced manufacturing base in China, established affiliates in Sweden, the United States, India, and Italy, built an efficient professional globalization team, and delivered a variety of products and services to more than 40 countries and regions.
Forward-Looking Statements
The information in this press release may include some forward-looking statements. Such statements are essentially susceptible to considerable risks and uncertainties. The use of “predicted”, “believed”, “forecast”, “planned” and/or other similar words/phrases in all statements related to our company is to indicate that the statements are forward-looking ones. Our company undertakes no obligation to constantly revise such predicted statements.
Forward-looking statements are based on our company management’s current perspectives, assumptions, expectations, estimations, predictions and understanding of future affairs at the time of the making of such statements. Such statements are not guaranties of future development and are susceptible to the impact of risks, uncertainties and other factors; some are beyond the control of our company and unpredictable. Subject to the influence of future changes and development in our business, competition environment, political, economic, legal and social conditions, the actual outcomes may differ significantly from the information contained in the forward-looking statements.












